Choosing the Right Reference Electrode
In electrochemical testing, attention is often placed on the working electrode, catalyst design, or electrolyte composition.
The reference electrode, by comparison, is frequently treated as a fixed component — something standard, interchangeable, and rarely questioned.
In reality, this assumption can introduce subtle but significant errors. An inappropriate reference electrode does not just shift your measured potential — it can affect stability, reproducibility, and even the interpretation of your results.
Why Reference Electrodes Matter More Than You Think
A reference electrode defines the potential scale of your entire experiment.
Every reported voltage — whether for reaction onset, redox peaks, or catalytic activity — is measured relative to this reference.
This means:
• A drift in reference potential → directly affects your data
• A mismatch between system and electrode → introduces hidden errors
• Improper selection → leads to misleading comparisons across studies
In other words, the reference electrode is not just a supporting component — it is the anchor of your measurement system.
A Common Oversight in Lab Practice
In many labs, one reference electrode (often Ag/AgCl) is used across different systems by default.
While convenient, this approach overlooks an important reality:
Not all electrochemical environments are compatible with the same reference electrode.
Factors such as:
• pH (acidic vs alkaline)
• presence of chloride ions
• temperature conditions
• solvent system (aqueous vs non-aqueous)
can all influence whether a reference electrode remains stable — or introduces error.
Understanding Common Reference Electrodes
Rather than viewing reference electrodes as interchangeable, it is more useful to understand their working environments and limitations.
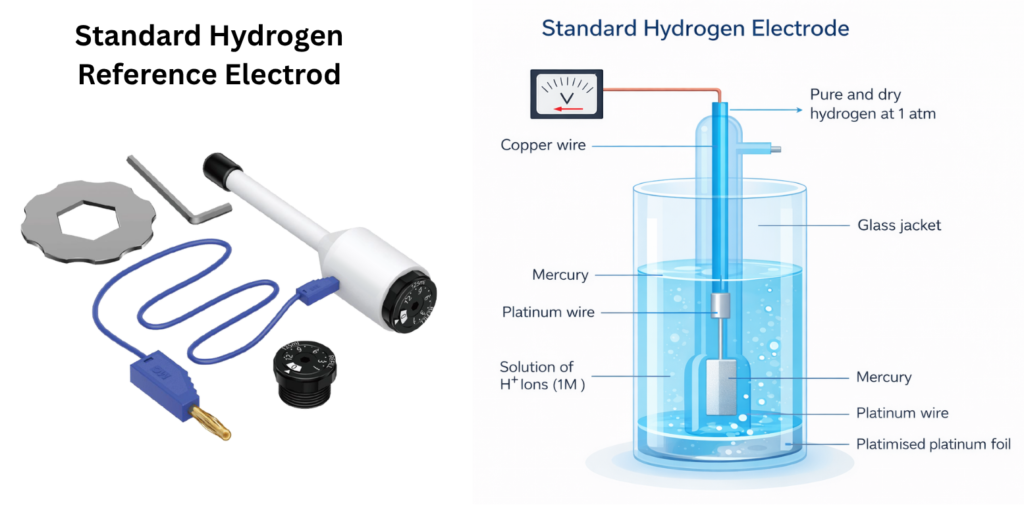
The Standard Hydrogen Electrode serves as the absolute reference point for all electrode potentials, defined as 0.000 V at any temperature.
However, due to:
• continuous hydrogen gas supply requirements
• safety concerns
• susceptibility to poisoning
it is rarely used in routine laboratory work, and is mainly reserved for calibration and theoretical studies.
2.Saturated Calomel Electrode (SCE)
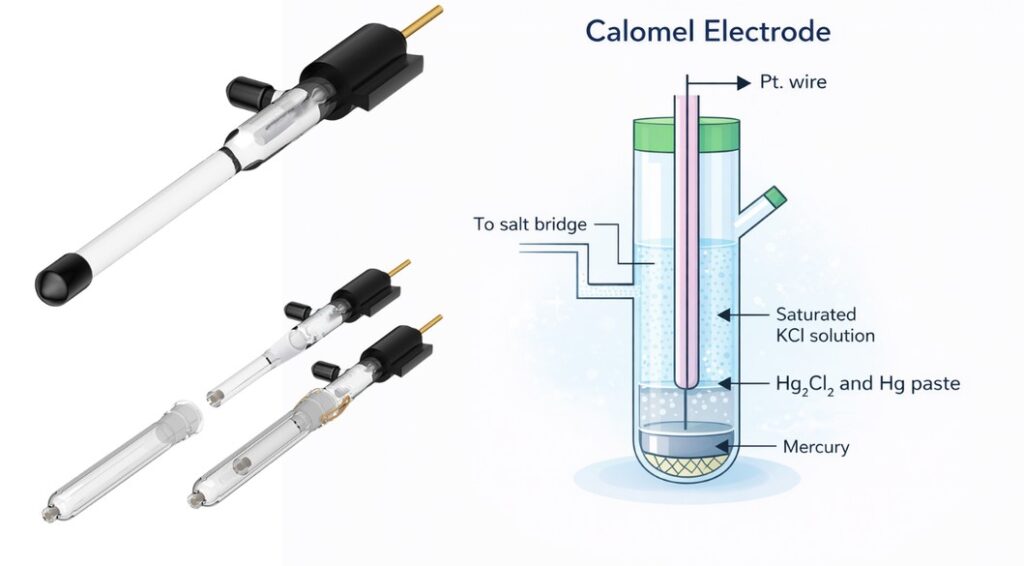
The Saturated Calomel Electrode is known for its:
- highly stable potential
- excellent reproducibility
At 25°C, its potential is approximately +0.241 V vs SHE.
However, practical limitations include:
- mercury toxicity
- instability at elevated temperatures (>80°C)
- chloride ion presence
As a result, its use has been gradually reduced in many modern laboratories.
3. Silver/Silver Chloride Electrode (Ag/AgCl)
Today, the Ag/AgCl electrode is one of the most widely used reference electrodes.

It offers:
• simple preparation
• good stability
• non-toxic composition
At 25°C (saturated KCl), its potential is approximately +0.197 V vs SHE.
It is well suited for most aqueous systems, and can also be adapted for non-aqueous environments by modifying the internal electrolyte.
4.Mercury/Mercury Oxide Electrode (Hg/HgO)

For alkaline systems, the Hg/HgO electrode provides a more appropriate reference.
Its advantages include:
- excellent stability in alkaline media
- reduced liquid junction potential
At 25°C (1 M KOH), its potential is approximately +0.098 V vs SHE.
This makes it particularly suitable for applications such as:
- oxygen evolution reaction (OER)
- hydrogen evolution reaction (HER) in alkaline electrolytes
5.Mercury/Mercurous Sulfate Electrode (Hg/Hg₂SO₄)

In systems where chloride ions must be avoided, the Hg/Hg₂SO₄ electrode becomes a valuable alternative.
At 25°C, its potential is approximately +0.615 V vs SHE.
It is typically used in:
• acidic sulfate environments
• systems sensitive to chloride contamination
This is especially important when studying metals such as silver or lead, where chloride ions can interfere with electrochemical behavior.
Choosing the Right Reference Electrode
Instead of defaulting to a familiar option, a more reliable approach is to match the electrode to your system:
- Acidic or neutral aqueous systems → Ag/AgCl or SCE
- Alkaline systems → Hg/HgO
- Chloride-sensitive systems → Hg/Hg₂SO₄
- Calibration / theoretical work → SHE
The key is not which electrode is “best,” but which is most appropriate for your testing environment.
Final Thoughts
In electrochemical experiments, accuracy is often thought to depend on advanced instrumentation or material design.
Yet in many cases, small foundational choices — such as the reference electrode — play an equally critical role.
By understanding the characteristics and limitations of different reference electrodes, labs can improve not only measurement reliability, but also confidence in their results.
Other Blog Posts You Might Like
Case Study: Relocation Solution for Electrochemical Equipment
Project Overview A research institution faced the daunting task of relocating two vital laboratory furnaces.…
Read moreKnowledge Education: Choosing the Right Reference Electrodes for Your Research
A reference electrode is used as a benchmark to measure the potential difference relative to…
Read moreElectrocatalysis: A Key Technology for Energy Storage and Conversion
As the world transitions towards renewable energy, electrocatalysis has become a crucial technology for energy…
Read more


